 Si (s) + 2 Cl 2 (g) → SiCl 4 (s) (Silicon (Vl) chloride) Si (s) + 2 F 2 (g) → SiF 4 (s) (Silicon (Vl) fluoride) The metal reacts with all Halogens to form Silicon (Vl) halides: If silicon is heated above 1400 o C, it reacts with the nitrogen, and forming Silicon nitrides: If silicon is heated above 900 o C, it reacts with the oxygen, and forming Silicon dioxide: Silicon doesn’t react with Air under normal condition: Isotopes: 28 Si 29 Si 30 Si 31 Si 32 Si Isotope Lattice constant: 543.07, 543.07, 543.07 pmįace Centered Diamond Cubic (FCDC) Reactivity of SiliconĮlectron affinity: 133.6 kJ/mol Nuclear Properties of Silicon Ionization energies: 1st: 786.4 kJ.mol 2nd: 1577 kJ/mol 3rd: 3231.5 kJ/molĬrystal structure: Face centered diamond cubic Sound Speed: 8433 m/s Atomic Properties of Silicon Molar magnetic susceptibility: -0045×10 -9 m 3/mol Physical Properties of Siliconĭensity: 2.33 g/cm 3 (In solid) 2.57 g/cm 3 (In Liquid at M.P) Mass magnetic susceptibility: -1.6×10 -9 m 3/kg Volume magnetic susceptibility: -0.00000373 Magnetic susceptibility (x mol): -3.9×10 -6 cm 3/mol Thermal conductivity: 149 W/(m∙K) Electrical properties of Siliconīand gap: 1.12 eV Magnetic Properties of Silicon When a very small cation combines with a very large anion, the resulting compound is less likely to exhibit the characteristic macroscopic properties of an ionic substance.Silicon Electron Configuration Thermal Properties of Siliconĭebye temperature: 645 K (701.33 oC, 371.85 oF) The larger the anion, the farther it is from the sodium ion, and the weaker the coulombic force of attraction between them. The sizes of singly charged cations, for example, increase in the following order: Li + NaCl (808☌) > NaBr (750☌) > NaI (662☌). 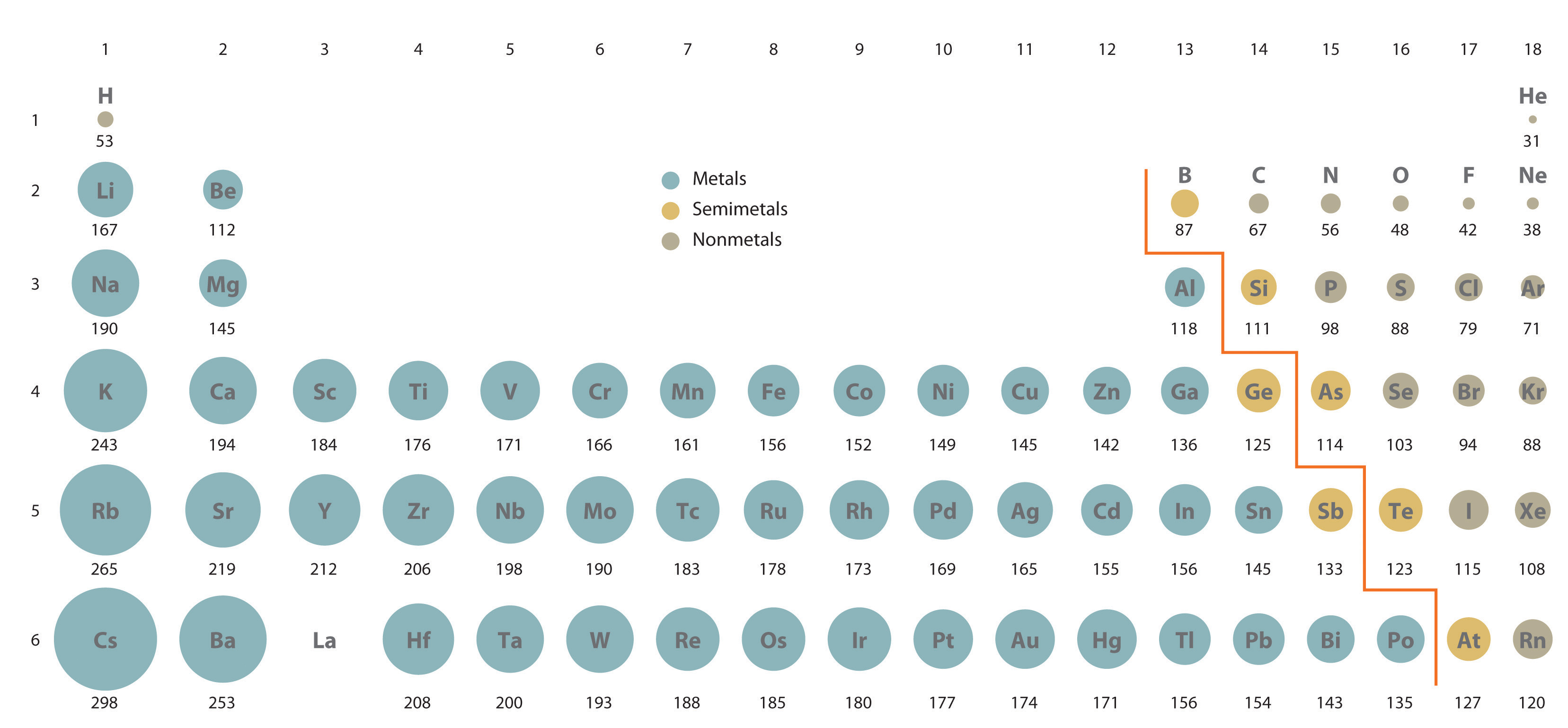
If we move down any of the columns, ionic sizes increase due to the increasing principal quantum number of the outermost electrons. Each row in the figure corresponds to an isoelectronic series involving a different noble-gas electron configuration.Īs we move from the more negative to the more positive ions in each row, there is a steady decrease in size. For any electronic series, such as H –, He, Li +, Be 2 +, in which the nuclear charge increases by 1 each time, we find a progressive decrease in size due to the increasingly strong attraction of the nucleus for the electron cloud. Species which have the same electronic structure but different charges are said to be isoelectronic. As we move across each row, the radius of each circle decreases. The final row has T e superscript 2 negative to T l superscript 3 positive. Fourth row has S e superscript 2 negative to I n superscript 3 positive. Third row has S superscript 2 negative to G a superscript 3 positive. The second row is N superscript 3 negative to Al superscript 3 positive.  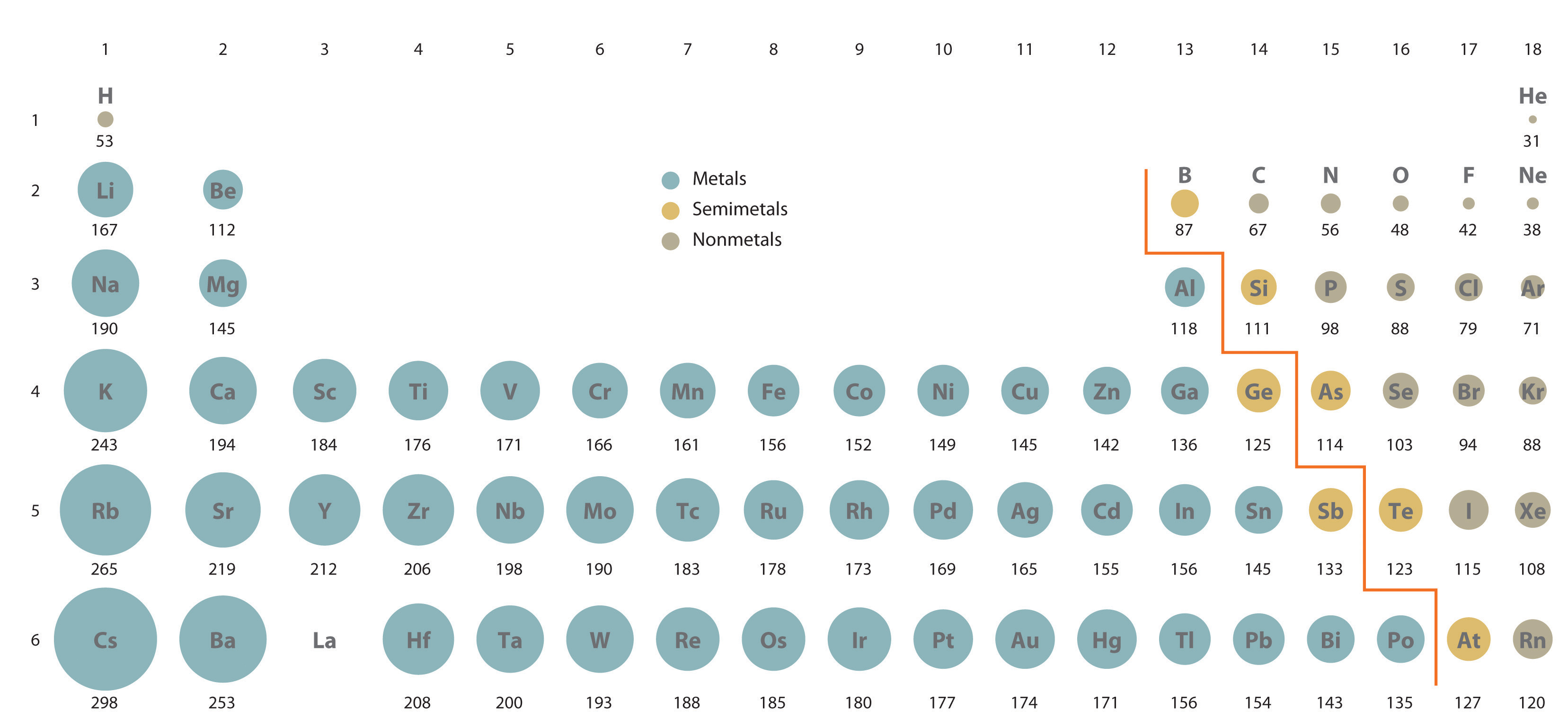
The first row is H negative to B e superscript 2 positive. Each row consists of elements as well as ions which have the same number of electrons. The elements of the periodic table are shown as circles with different radius. Ions in the first row of this figure, H –, Li +, and Be 2 +, all have the same 1 s 2 electronic structure as the helium (He) atom, but differ in size due to the different number of protons each has in their nucleus. This relationship is evident in the following figure comparing ionic radii. The size of an ion is governed not only by its electronic structure but also by its charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
